Call: 08045804253
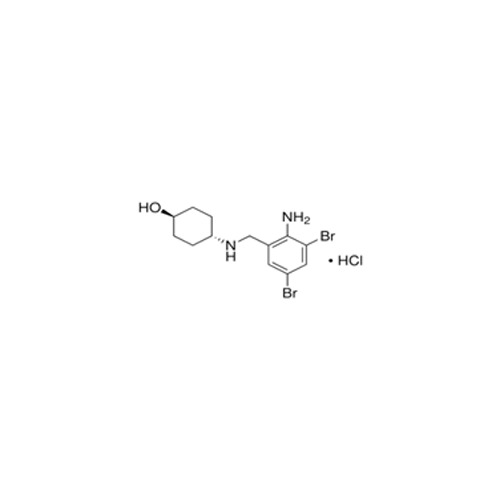
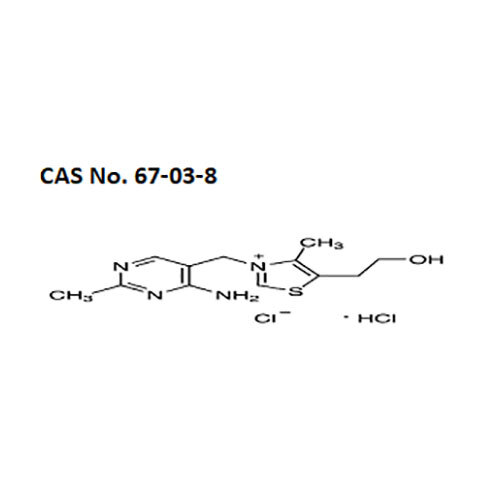
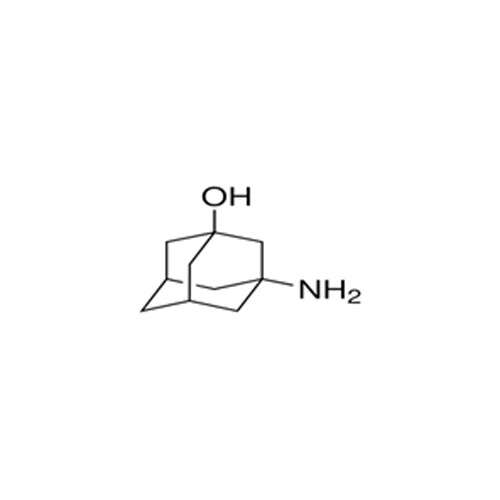
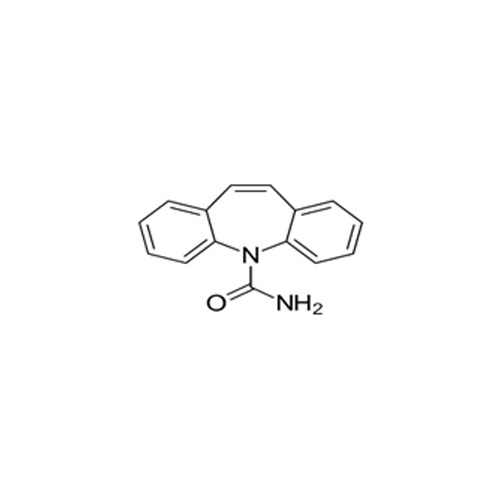
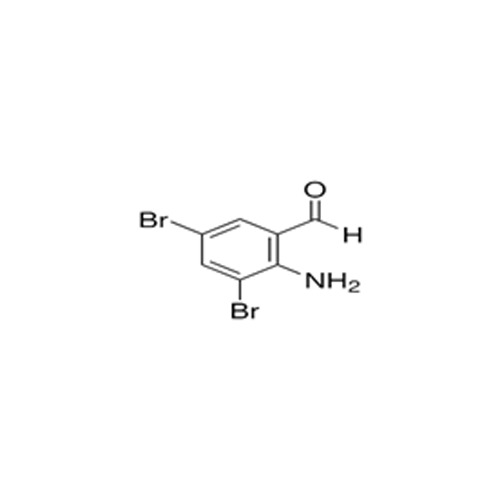
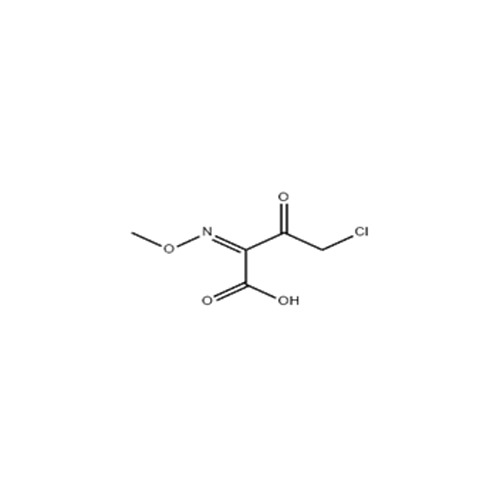
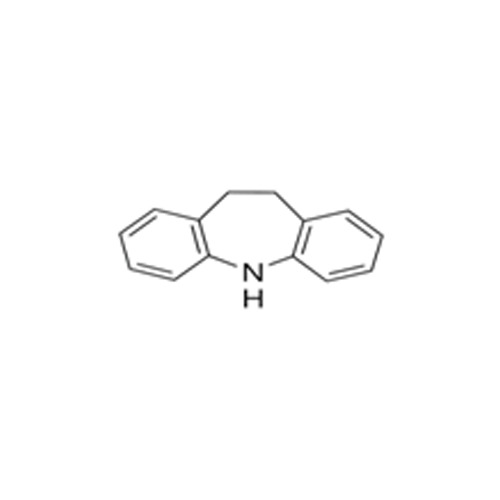
High quality Pharma Intermediates such as 4-(Chloromethyl) Benzoic Acid, Iminodibenzyl (IDB), ISBCC, 10-Methoxy iminostilbene (10-MIS), 2-amino-3, 5 dibromobenzaldehyde (ADBA), Trans-4-aminocyclohexanol (4-TACH) and new intermediates with cost effective RoS for driving innovation and growth in Pharmaceutical Industry.
- New Arrivals
- Featured
Escogen Lifesciences Pvt. Ltd. incorporated in the year 2022 under Companies Act 2013, is one of the fast-growing pharmaceutical companies engaged in manufacturing and supply of APIs and Intermediates. The facility has capabilities for performing simple to complex reactions to give quality products on committed time. The Regulatory Guidelines and Environment, Health and Safety (EHS) are the primary considerations in the organization. Our business model focuses on earning trust, maintain transparency and share strong bond of togetherness with our customers and partners. At Escogen Lifesciences, we are committed to work continuously with innovative ways to improve our processes, quality, EHS, compliances and cost effectiveness.
Escogen Lifesciences has strong capabilities in chemistry and infrastructure to supply complex intermediates. The patented products are manufactured only for R&D and studies purposes permitted under the law. The company doesnt offer any commercial supply to the countries where any patent is in force or anticipated to have intellectual property infringement or any other regulatory issue.
Facilities
The plants are equipped with SS reactors of 1 KL to 6 KL capacity, Glass lines reactors from 1 KL to 6KL capacity, centrifuges of 36/48/60 inches, various types of dryers (like vacuum dryers, FBD and Air driers) to manufacture the products as per GMP guidelines. Appropriate size compressors and cooling towers for cooling/chilling, boiler for steam, compressed air. nitrogen for inertization, purified water, air handling units, and other utilities are provided to carry out the required reactions. Storage of raw materials, intermediates and APIs are done as per product requirement following the GMP guidelines.
The analytical facility is equipped with most advanced analytical equipment to carry out analysis as per specifications.
The company is developing advanced Research & Development Facility to carry out complex chemical and biotech reactions. The R&D focuses continuously to introduce new products, to improve quality and to reduce the cost with specific focus on Environment, Health and Safety in processes and products. The storage facility is provided as per requirement of the products which includes Cold Storage, controlled temperature and ambient as per requirement of the products. An efficient Effluent Treatment Plant is installed to treat liquid effluent to meet the required norms of the liquid discharge.
Services
We also offer consultancy services in contract research, technical and management area. The consultancy services include:
Management Team
The management team is well qualified and experienced in Pharma Industry having long experience in Manufacturing, R&D, Supply Chain, GMPs, EHS, compliances and systems development.
Escogen Lifesciences has strong capabilities in chemistry and infrastructure to supply complex intermediates. The patented products are manufactured only for R&D and studies purposes permitted under the law. The company doesnt offer any commercial supply to the countries where any patent is in force or anticipated to have intellectual property infringement or any other regulatory issue.
Facilities
The plants are equipped with SS reactors of 1 KL to 6 KL capacity, Glass lines reactors from 1 KL to 6KL capacity, centrifuges of 36/48/60 inches, various types of dryers (like vacuum dryers, FBD and Air driers) to manufacture the products as per GMP guidelines. Appropriate size compressors and cooling towers for cooling/chilling, boiler for steam, compressed air. nitrogen for inertization, purified water, air handling units, and other utilities are provided to carry out the required reactions. Storage of raw materials, intermediates and APIs are done as per product requirement following the GMP guidelines.
The analytical facility is equipped with most advanced analytical equipment to carry out analysis as per specifications.
The company is developing advanced Research & Development Facility to carry out complex chemical and biotech reactions. The R&D focuses continuously to introduce new products, to improve quality and to reduce the cost with specific focus on Environment, Health and Safety in processes and products. The storage facility is provided as per requirement of the products which includes Cold Storage, controlled temperature and ambient as per requirement of the products. An efficient Effluent Treatment Plant is installed to treat liquid effluent to meet the required norms of the liquid discharge.
Services
We also offer consultancy services in contract research, technical and management area. The consultancy services include:
- Process development and Optimization at commercial stage.
- New Product Development from design to commercialization.
- Patent screening of new products and provide alternate ROS
- Quality Improvement with cost reduction.
- Plant design as per cGMP Guidelines
- Optimization of supply chain.
- MIS design and implementation.
- System audits for Quality and EHS
- Manpower studies and optimization.
Management Team
The management team is well qualified and experienced in Pharma Industry having long experience in Manufacturing, R&D, Supply Chain, GMPs, EHS, compliances and systems development.
Back to top

 Send Inquiry
Send Inquiry




![Dibenzo[b.f][1,4]thiazepin-11 (10H)-1-one Dibenzo[b.f][1,4]thiazepin-11 (10H)-1-one](https://cpimg.tistatic.com/11292838/b/4/Dibenzo-b-f-1-4-thiazepin-11-10H-1-one..png)